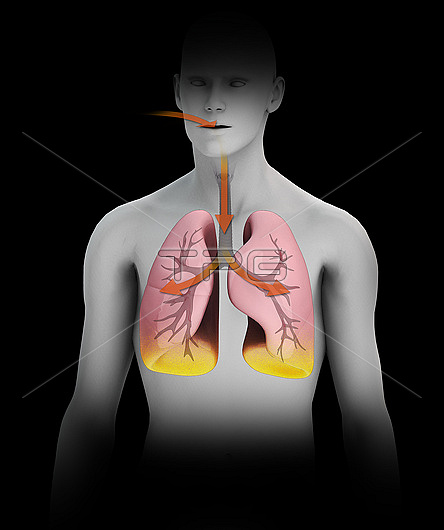
Radon decaying to polonium in humans, illustration. Radon is a chemical element that is a radioactive gas that occurs in nature. It is being continuously generated from radioactive isotopes in the Earth's crust. The most stable isotope of radon is radon-222, which has a half-life of 3.8 days. As a gas, it can be inhaled into the lungs where it decays via alpha particle production to produce polonium-218, a solid that has a half-life of just over three minutes. Further decay takes places, eventually producing a stable isotope of lead. The effects of this can lead to an increased risk of lung cancer. For this illustration with labels, see image C047/4523.
| px | px | dpi | = | cm | x | cm | = | MB |
Details
Creative#:
TOP26514937
Source:
達志影像
Authorization Type:
RM
Release Information:
須由TPG 完整授權
Model Release:
N/A
Property Release:
N/A
Right to Privacy:
No
Same folder images:

 Loading
Loading